
The BrF3 molecule has 28 valence electrons. It needs one more electron to get a stable octet noble gas configuration. In their outermost shells, both atoms have seven electrons. They both fall in the same halogen family. Fluorine and bromine atoms have seven valence electrons. The core central atom of BrF3 molecule is bromine, which is surrounded by three fluorine atoms. calculation of molecular mass of BrF3īromine trifluoride(BrF3) has a chemical composition of one bromine central atom and three fluorine atoms in that molecular geometry. 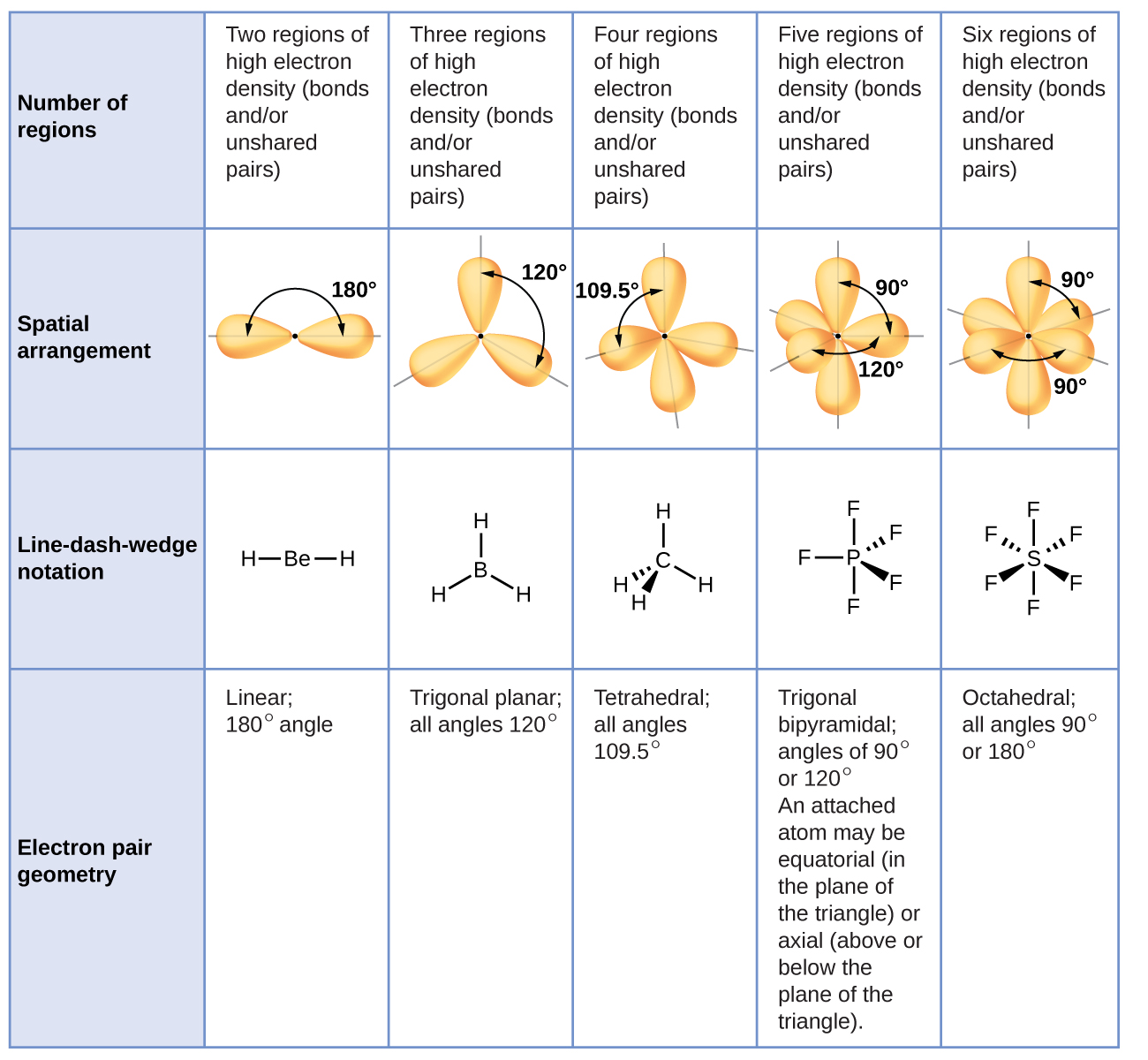
Molecular mass of BrF3 = 1 * 79.9 (atomic mass of Br) + 3 * 18.9 (atomic mass of F) = 136.90 g/mol. SbF3 + BrF3 -–> SbBrF6 formation of polyhalides Properties of BrF3īrF3 molecule has a molecular mass of 136.90 g/mol. another one polyhalides preparation reaction is as follows Bromine trifluoride (BrF3) reacted with tungsten oxide, forms tungsten hexafluoride, bromine, and oxygen.īrF3 reacted with salt: potassium fluoride (KF) salt reacted with bromine trifluoride and forms polyhalides.īrF3 + KF -> KBrF4 formation of polyhalidesīromine trifluoride used to prepare polyhalides. The chemical formula for tungsten oxide is WO3. Reaction with tungsten oxide(WO3): Tungsten is a metal. Bromine trifluoride (BrF3) reacted with silica(SiO2), forms silicon tetrafluoride, bromine, and oxygen. It is generally called the metalloid family. silicon element comes under nonmetallic family. Reaction with Silica(SiO2): Silica (SiO2) is not metallic oxide. But, Bromine trifluoride is a very reactive substance and reacted well with the oxides materials also. 
It won’t undergo any chemical reactions easily. Normally all the oxides of metal and nonmetal are stable compounds. The reaction of BrF3 with waster as followsīrF3 + 3H2O -–> 3HF + H3BrO3 Hydrolysis reaction of bromine trifluoride(BrF3)īromine trifluoride reacted with water and gives hydrogen fluoride(HF) and H3BrO3 acid.īrF3 reacted with metal and nonmettalic acompounds as followsĤBrF3 + 3SiO2 -–> 3SiF4 + 2Br2 + 3O2 BrF3 reacted with silica(SiO2)- nonmetallic compoundĤBrF3 + 2WO3 -–> 2WF6 +2Br2 +3O2 BrF3 reacted with tungsten oxide (WO3)- metallic compound The order of chemical reactivity is as followsĬlF3 > BrF3>IF7>BrF5>IF5>BrF Order of chemical reactivity of interhalogen compounds It is one of the highly reactive interhalogen compound. Bromine vapor reacted with fluorine gas diluted with nitrogen gas gives bromine trifluoride(BrF3)īr2(vapour) + 3F2 (diluted with N2) -–> 2BrF3 The formation of BrF3 It was made by combining bromine vapor and fluorine at the temperature of 20☌. Paul Lebeau, was a well-known French Chemist, was the first to discover it in 1906. External References: “Is BrF3 polar or nonpolar ?”.BrF3 Polarity, Molecular geometry, Hybridization, and Bond angle.BrF3 synthesis and chemical reactivity:.Due to the existence of very reactive three fluorine atoms linked together, it is a potent fluorinating agent in an organic chemistry laboratory. When it comes into contact with water or other organic materials, it reacts quickly. It exists in a straw color liquid with a strong pungent odor at standard temperature and pressure conditions. Interhalogen bromine trifluoride (BrF3) is a chemical with three fluorine atoms and a central bromine atom. And because the charge distribution between bromine and three fluorine atoms in BrF3 molecule isn’t uniform, the BrF3 molecule is polar. Is BrF3 a polar or nonpolar molecule in nature? Because the presence of two lone pairs on the bromine atom causes the molecule’s form to be deformed or bent, BrF3 (bromine trifluoride) is a polar molecule. In this blog post, we are going to discuss these in a detailed manner.īrF3 has a pungent odor and appearance as straw colored liquid. The student used to ask “Is BrF3 polar or nonpolar?”, “BrF3 Lewis structure”, “BrF3 molecular geometry”, and “polarity of BrF3”. All four atoms in this BrF3 molecule have high electronegativity values. It contains one bromine and three fluorine atoms. Bromine trifluoride has the chemical formula BrF3 and interhalogen compound.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
